Fundamentals of Batteries
Batteries can be divided into two categories. The primary type is intended for one time use only and is disposed after the charge has dropped to a level that cannot be used. Primary type should not be discharged as heat will be generated within sealed cells.
It will also damage the equipment as a consequent of fluid leakage. The storage or secondary type can be recharged many times and is reusable.
The rating of its capacity is ampere hours (Ah) which is a product of current drain and time.
If it become cold, it will have less charge available and some design to keep it warm before use. It may lose 70% of its capacity at cold extremes but will recover with warmth.
Primary Type
Carbon zinc is the most common primary cell in which the chemical oxidation converts the zinc into salts and electricity. When there is no current flowing, the oxidation stops. If keep for a long period of time, the stored cell will degrade and dry out where it will no longer able to supply the desired current. The time taken for the degradation without being used is called shelf life. It has a nominal voltage of 1.5V.
Alkaline types have longer capacity at low temperatures. Lithium type have nominal voltage of 3V/cell and has the best capacity, discharge, shelf life and temperature characteristics. Its setback is the high cost.
Silver Oxide and Mercury has voltages of 1.5 V and 1.4 V respectively and are used where constant voltage is desired at low currents over a long period of time. Their main used and applications are in hearing aids.
Storage Type
The most common type is nickel-cadmium(Ni-Cd) type with a nominal voltage of 1.2V/cell. If used carefully, it can be rechargeable up to 500 times compared to alkaline type which is 50 times or so.
The most widely used storage type is the lead-acid type in automobile.The Lead Acid battery is made up of plates, lead, and lead oxide with a 35% sulfuric acid and 65% water solution.
Gas escaping from it may be explosive and always keep flame away. It should not be subjected to unnecessary heat, vibration or physical shock. Frequent inspections for leak is recommended. The electrolyte is chemically active and conductive and may ruin electrical equipment if leaks occurred. Its acidity may be neutralized with sodium bicarbonate or baking soda.
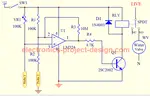
In order to ensure that all the cells in NiCd reach a fully charged condition, it should be charged by a constant current of 0.1 C current level. It is around 50 mA for a AA size cells. Charging should be terminated after 15 hours at the slow rate. A built in circuit that will stop charging when 1.43V/cell is reached will enhance the life of the battery.
New! Comments
Have your say about what you just read! Leave us a comment in the box below.